Redox flow batteries are promising candidates for grid-scale electrical energy storage, mainly due to their ability to scale power and energy capacity independently (Figure 1). Widespread utilisation is currently impeded by high system costs, owing partly to the use of expensive electrolytes. A price reduction could be achieved by using cheap organic compounds as active materials for aqueous electrolytes.
The aims of this PhD project are to:
• Screen a large group of substituted quinone and anthraquinone species to determine their aqueous redox potentials and solubilities, among others (Figure 2).
• Carry out single cell tests with the most promising compounds as anolyte and catholyte active materials via cell cycling and electrochemical impedance spectroscopy protocols.
• Establish a reliable equivalent circuit model for fitting the impedance response of aqueous organic redox flow batteries.
The project is partly funded by Innovation Fond Denmark through the grand solution project ”ORBATS".
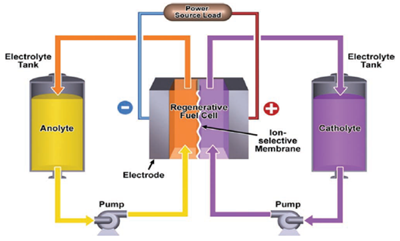
Figure 1. Operating principle of a redox flow battery.
[W. Wang et al, “Recent Progress in Redox Flow Battery Research and Development, ”Adv. Funct. Mater., vol. 23, no. 8, pp. 970–986, 2013.]

Figure 2. Solubility and redox potential of some organic redox-active species.
[R. Ye et al., “Redox Flow Batteries for Energy Storage: A Technology Review, ”J. Electrochem. En. Conv. Stor., vol. 15, no. 1, pp. 010801 1–21, 2017.]