Electrochemical CO2 reduction at high temperature in solid oxide electrolysis cells
 .
.
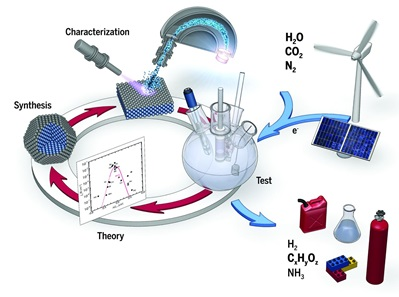
Image credit: Jakob Kibsgaard, DTU PhysicsThe highly extensive utilization of fossil fuels within the current energy infrastructure is regarded as one of the most intensive sources of carbon dioxide emissions, which is argued to be responsible for the increase of the greenhouse effect, resulting in global warming and climate changes. The present project aims to develop advanced solid catalysts for the electrochemical conversion of carbon dioxide to hydrocarbons and oxygenated organic compounds, which is considered a promising strategy for providing a long-term solution to mitigating global warming and energy supply problems. In this respect, ceria-based materials form a very promising class of electrocatalysts for carbon dioxide reduction reaction in high temperature solid oxide electrolysis cells (SOECs). The aim of the study is to gain better understanding of the reaction mechanisms and their relation to the ceria composition and state of its surface, by employing highly tailored thin-film hetero-structures prepared by pulsed laser deposition (PLD). The study will address the effect of different surface orientations and the role of tensile and compressive isotropic strain introduced in the lattice, as well as that of acceptor and donor dopants in different concentrations. The investigation of size/shape-dependent properties in size-tunable CeO2 nanocrystals will also be pursued. The systematic approach described earlier is anticipated to result in the fundamental understanding of the reaction mechanism, the steps that limit the reaction rate, and how these can be accelerated by appropriate tuning of the electrocatalyst material properties.