To combat anthropogenic climate change it is necessary to switch the world’s supply of energy away from CO2 polluting fuels. One such way is to use hydrogen as an energy carrier and green fuel. The production of hydrogen can be achieved by splitting water into oxygen and hydrogen. If any technology is to have an impact in slowing climate change it needs to be scaleable to the terawatt scale. This challenge precludes any technology reliant on very rare earth metals.
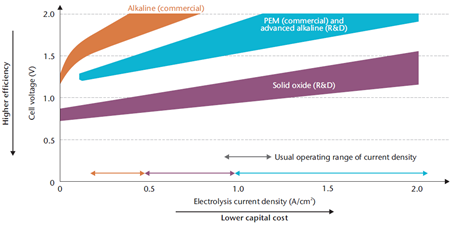
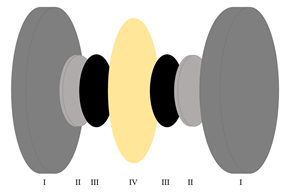
This PhD project aims at creating an alkaline electrolysis cell capable of drawing 2 A/cm2 at a cell voltage of 2 V. This will be attempted by utilizing catalyst layers either sprayed on electrode supports or by coating bulk materials in nano-sized catalyst. This novel architecture allows for the use of cell design that is closely packed, as to avoid electrical losses, and allows for the high surface area of nanoparticles that to eliminate activation losses.
If successful, this would allow for novel cell designs that mirror those of the PEM-electrolysers to harvest the non-noble metals of the alkaline system and the high performance of the acidic system.